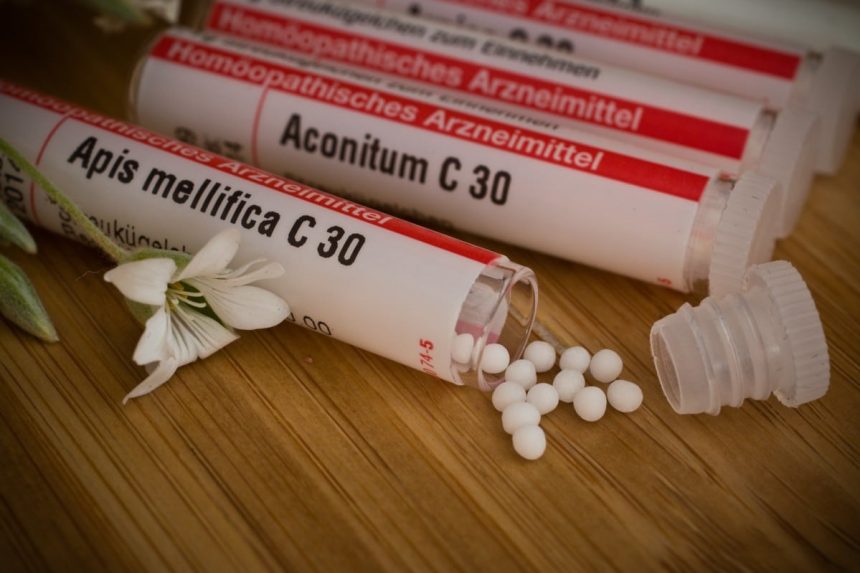
A leading European science federation that advises EU institutions is calling for a ban on the use of homeopathy by publicly funded health bodies and tough new controls on the marketing of homeopathic products.
In a new statement (published on 20 September), a Working Group of the European Academies’ Science Advisory Council (EASAC) says there is “no robust, reproducible evidence that homeopathic products are effective for any known diseases, even if there is sometimes a placebo effect”. EASAC argues that, in addition, homeopathy can be harmful by delaying or deterring patients from getting advice from a qualified medical practitioner, and that it “undermines patient and public confidence in scientific evidence”.
EASAC calls the scientific claims made for homeopathy “implausible” and “inconsistent” with established concepts from chemistry and physics.
“Evidence-based public health systems should not offer reimbursement for homeopathic products”
In its statement, EASAC says: “We recognize the fundamental importance of allowing and supporting consumer choice. But patient choice must be appropriately informed and this raises issues for achieving a standardised, knowledge-based, regulatory framework and sound advertising practices that can apply equitably to all medicinal products, whatever their origins and whatever their mechanisms.
The Statement goes on to make a series of recommendations:
• “There should be a consistent regulatory requirement for claims for the efficacy, safety and quality of all medicinal products to be based on verifiable and objective evidence, commensurate with the claims being made. The necessity for robust data applies to products for both human and veterinary medicine.
• Public health system budgets are under increasing pressure. Evidence-based public health systems should not offer reimbursement for homeopathic products and services unless they are demonstrated to be efficacious and safe by rigorous testing.
• The composition of homeopathic products should be labelled in a similar way to other health products available in the pharmacy (OTC) or elsewhere. That is, the current exceptional labelling permitted for homeopathic products should be replaced by a simple description of the ingredients and their amounts present in the formulation.
• Advertising and marketing of homeopathic products and services must be regulated to be accurate and clear: advertising claims made for efficacy and safety should not be allowed without demonstrable and reproducible evidence.”
EASAC says that its recommendations on testing, regulation, labelling and marketing of homeopathic products have “significant implications” for several EU institutions “and for patient education and dialogue, and public engagement”.